“Is Aluminium magnetic or non-magnetic?” this simple question sometimes confuses many professionals. Aluminum is one of the lightweight and widely used metals for industrial applications. To answer the question if Aluminum is magnetic, we have to explore more about its properties. Let’s dive into finding the solution to this widely asked question.
In its pure form, aluminum does not exhibit magnetic properties. It falls under the category of non-ferromagnetic materials, which do not possess a permanent magnetic field. When exposed to a magnetic field, aluminum does not attract or repel the magnet. However, under certain conditions, Aluminum can exhibit weakly magnetic behavior. Aluminum is a paramagnetic material and all paramagnetic materials can show weak magnetic properties under imposed magnetic fields. To learn this, let’s find out the electronic configuration of Aluminum.
Electronic Configuration of Aluminum
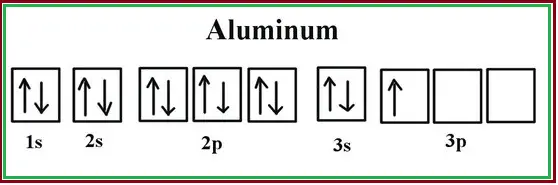
The electronic configuration of aluminum is 1s² 2s² 2p⁶ 3s² 3p¹. Let’s break down the electronic configuration of aluminum:
- 1s²: This indicates that there are two electrons in the 1s orbital. The 1s orbital is the innermost energy level or shell.
- 2s²: This signifies that there are two electrons in the 2s orbital. The 2s orbital is the second energy level or shell.
- 2p⁶: This indicates that there are six electrons in the 2p orbitals. The 2p orbitals belong to the second energy level or shell and can hold a maximum of six electrons.
- 3s²: This signifies that there are two electrons in the 3s orbital. The 3s orbital is the third energy level or shell.
- 3p¹: This indicates that there is one electron in the 3p orbital. The 3p orbital belongs to the third energy level or shell and can hold a maximum of six electrons.
In total, aluminum has 13 electrons, which occupy the various orbitals as described above. The electronic configuration provides information about how the electrons are distributed in the different energy levels and orbitals of an atom. The same can be found in Fig. 1 below.
Why does Aluminum show Weak Magnetic Behavior?
We all are aware that paired electrons spin in opposite directions which cancels out the net dipole moment. However, for unpaired electrons, during their spin, the net dipole moment does not cancel out and It makes them tiny magnets. The behavior will increase with an increase in the number of unpaired electrons. As we have seen in Fig. 1, aluminum has only one unpaired electron. Because of this, in the presence of an external magnetic field, Aluminum weakly attracts a magnet.
Note that when there is no externally imposed magnetic field, the magnetic behavior of aluminum or other paramagnetic materials is lost as they can not retain magnetization similar to ferromagnetic materials.
How to Induce External Magnetic Field for Aluminum?
An external magnetic field can easily be induced by moving the aluminum near a magnet. That is the reason, sometimes, you may find an aluminum can to move or float slowly when passing through a hanging magnet.
So, the above discussion clearly answers the question “Is Aluminum Magnetic?”. The direct answer is Pure aluminum is non-magnetic under normal circumstances but when an external magnetic field is imposed, it shows a weak magnetic property.
Factors Influencing Aluminum’s Magnetism
Several factors contribute to the magnetic behavior of aluminum:
Electron Configuration:
The atomic structure of aluminum involves three valence electrons. These electrons are relatively loosely bound and can move freely within the metal lattice. However, their movement does not result in the establishment of a permanent magnetic field, unlike in ferromagnetic materials.
Crystal Structure:
Aluminum has a face-centered cubic crystal structure. This arrangement of atoms within the material does not allow for the alignment of magnetic domains, which is necessary for the establishment of magnetism.
Magnetic Susceptibility:
Aluminum has a very low magnetic susceptibility (2.2 χm), indicating its weak response to an applied magnetic field. The low magnetic susceptibility arises from the absence of unpaired electrons or magnetic moments within the aluminum atoms.
Misconceptions and Confusions
Confusion regarding aluminum’s magnetism often arises due to misconceptions or the presence of magnetic coatings on aluminum surfaces. It is important to differentiate between the intrinsic properties of aluminum and the influence of external factors.
Magnetic Coatings:
Aluminum objects may be coated with magnetic materials, such as iron oxide or nickel, for specific purposes or applications. These coatings can give the appearance of magnetism, but it is important to note that the underlying aluminum material itself remains non-magnetic.
Induced Currents:
When exposed to a changing magnetic field, aluminum can experience electromagnetic induction, resulting in the generation of electric currents. These induced currents can exhibit a magnetic effect, known as an eddy current. However, this effect is temporary and disappears once the magnetic field is removed.
Practical Applications
Aluminum’s non-magnetic nature offers distinct advantages in various applications:
- Electrical and Electronics: Aluminum’s low magnetic susceptibility makes it suitable for use in electrical conductors and components where magnetic interference or eddy currents need to be minimized.
- Magnetic Shielding: Aluminum’s non-magnetic property can be utilized for magnetic shielding applications, where it acts as a barrier to redirect or block magnetic fields from sensitive equipment or areas.
- Non-Magnetic Tools and Equipment: Aluminum’s non-magnetic nature makes it ideal for manufacturing non-magnetic tools, equipment, and instruments used in industries such as healthcare, research, and electronics.
Conclusion
Aluminum, while an incredibly useful and versatile metal, is not magnetic in its pure form. Its non-ferromagnetic properties can be attributed to factors such as electron configuration, crystal structure, and low magnetic susceptibility. Understanding aluminum’s non-magnetic behavior is essential to utilize its unique properties effectively in various industries. While aluminum objects may exhibit magnetic effects due to coatings or induced currents, it is important to differentiate between the intrinsic non-magnetic nature of aluminum and external influences.
