“What is viscosity?” is an often-asked term in fluid mechanics. Viscosity is a fluid property and is very important for studying fluid flow behavior. All kinds of fluids, whether they are in a liquid state or gaseous state, possess viscosity. Viscosity is a fundamental property of fluids that plays a crucial role in various fields, including physics, engineering, chemistry, and even biology. It describes a fluid’s resistance to deformation and flow, which makes it essential for understanding the behavior of liquids and gases in both natural and industrial processes. In this article, we will explore more details about the term “viscosity,” its definition, significance, units, equations, and values.
Definition of Viscosity
The term “viscosity” has its root in the Latin term “viscum,” which refers to a viscous glue originating from mistletoe berries. In fluid mechanics, viscosity is defined as the measure of a fluid’s resistance to fluid flow under an applied force. It can be intuitively understood as the “thickness” or “stickiness” of a fluid. For a fluid in motion, viscosity describes internal friction. So, a fluid having a large viscosity provides more internal friction to resist flow, whereas a fluid with a lower viscosity provides little friction. In general, liquids have more viscosity than gases.
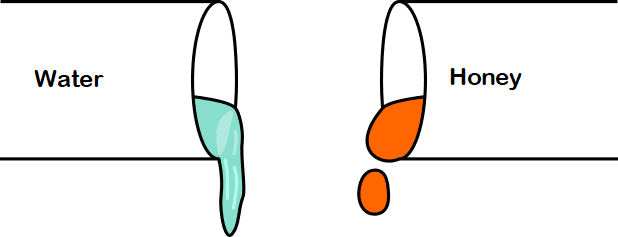
The concept of viscosity will be more clear if we consider the following example. If we take water and honey in a pot and try to pour, we find that water is flowing more smoothly and quicker than honey. This is because honey is more viscous than water. So, honey provides more resistance to motion than water, and that is why water moves more freely than honey.
Significance of Viscosity
The viscosity of a fluid is opposite to the fluidity that denotes how easily a fluid can flow. It is basically the internal friction between the molecules comprising the fluid. For fluid transportation and lubrication engineering, injection molding, spraying, and surface coating applications, viscosity plays a major role as it controls the flow of the liquid. Knowing the viscosity data is very important to predict fluid behavior. For example, if the tomato ketchup inside the tube does not have the correct viscosity, it may not flow from the tube or flow too much, all of certain.
Understanding viscosity is vital in a range of disciplines:
- In engineering, it influences the design of pipelines and pumps.
- In food science, it affects texture and mouthfeel.
- In pharmaceuticals, it determines how drugs are delivered.
Symbol of Viscosity
Mathematically, viscosity can be defined as the ratio of viscous stress (shear stress) to the rate of change of deformation. The symbol of viscosity is µ (Greek letter mu). Hence,
Viscosity, µ=Shear Stress/strain rate=τ / (du/dy)
So, µ=τ / (du/dy)……(1)
The above symbol for viscosity µ is widely used. However, some physicists and chemists prefer to use η (the Greek letter eta) as the symbol of viscosity.
Units of Viscosity
From the above mathematical equation, we know that viscosity = stress/strain rate. The unit of stress in SI unit =N/m2. The unit of Strain rate = (m/s)/m.=1/s
Hence, the unit of viscosity = (N/m2)/(1/s)=N-s/m2, and the dimension of viscosity is (force X time/area).
The above viscosity discussed is also popular as dynamic viscosity or absolute viscosity.
Hence, the unit of viscosity or dynamic viscosity in the SI system is N-s/m2 or pascal-second.
Often, the unit of viscosity is denoted by Poise or Centipoise. In the CGS unit system, the unit of dynamic viscosity is “Poise” named after Jean Léonard Marie Poiseuille. The relation between Pascal-Second and poise is given below:
1 Pascal-Second= 10 Poise or 1Pa-s=10P
Types of Viscosity
There are two types of viscosity, relevant in different contexts.
Dynamic (Absolute) Viscosity
Dynamic viscosity, often denoted by the symbol μ (mu), is a measure of a fluid’s internal resistance to flow when an external force is applied. It is expressed in units of Pascal-seconds (Pa·s) or poise (P), where 1 P = 0.1 Pa·s.
Kinematic Viscosity
Kinematic viscosity, represented by the symbol ν (nu), measures the fluid’s resistance to flow under gravity. It is defined as the ratio of dynamic viscosity to fluid density and is usually expressed in square meters per second (m²/s) or centistokes (cSt).
What is Kinematic Viscosity?
Kinematic viscosity or momentum diffusivity is defined as the ratio of dynamic viscosity to fluid density. The symbol of kinematic viscosity is ν (Greek letter nu). So, mathematically the formula for kinematic viscosity is given by ν=µ/ρ.
Now we just learned that the unit of dynamic viscosity=N-s/m2. Unit of fluid density=Kg/m3. Hence, the unit of kinematic viscosity =(N-s/m2)/(Kg/m3)=(Kg-m/s2)*(s/m2)*(m3/Kg)=m2/s.
Accordingly, the dimension of kinematic viscosity is (length2/time). In fluid dynamics, working with kinematic viscosity is more convenient.
Measuring Viscosity
It is well known that measuring the viscosity of fluids is very important to understand the flow characteristics of those fluids. There are various types of instruments by which viscosity can be measured. Those viscosity-measuring devices are known as viscometers and rheometers. Common, widely used instruments for measuring viscosity are:
- Capillary Viscometer
- Falling Sphere Viscometer
- Vibrating Viscometer
- Rotational Viscometer
- Microfluidic Rheometers
- Zahn Cup
- Fluorescence correlation spectroscopy
- Acoustic rheometer
Factors Affecting Viscosity
There are various factors that affect the viscosity of a fluid. Those are:
- Fluid Temperature: Usually the viscosity of liquids decreases with an increase in temperature. On the contrary, the viscosity of gases increases with an increase in temperature.
- Flow Conditions: For laminar flow, the viscosity of liquid remains constant while for turbulent flow viscosity changes.
- Pressure: With an increase in pressure, the viscosity of gases usually increases. Liquids being incompressible does not have much impact.
- Multiphase flow: The viscosity of multiphase flow is affected by the volume of each phase.
- Suspended Particles: Suspended materials increase viscosity.
- Shear Rate: In non-Newtonian fluids, viscosity is dependent on shear rate. For example, shear-thinning fluids decrease in viscosity with increased shear rate, while shear-thickening fluids increase in viscosity under the same conditions.
Viscosity Measurement Methods
Measuring viscosity is essential for both theoretical and practical applications. Several methods exist, each with its advantages and disadvantages.
Capillary Viscometers
Capillary viscometers operate by allowing a fluid to flow through a narrow tube. The time taken for a certain volume of fluid to pass through the tube is measured, allowing for the calculation of viscosity. These are particularly effective for low-viscosity fluids.
Rotational Viscometers
Rotational viscometers measure the torque required to rotate a spindle immersed in the fluid. The viscosity is derived from the relationship between torque and rotational speed. This method is suitable for a wide range of viscosities and provides real-time data.
Falling Sphere Viscometers
This method involves dropping a sphere through the fluid and measuring the time it takes to fall a certain distance. The viscosity can be calculated based on the sphere’s terminal velocity, density of the sphere, and the fluid’s density.
Oscillatory Viscometers
Oscillatory viscometers use a vibrating element to measure the fluid’s resistance to oscillatory motion. This method is particularly useful for non-Newtonian fluids and provides information about both viscosity and elasticity.
Viscosity of Water
The viscosity of water at 200 C is 1 centipoise or 1 cP. As for liquids, the viscosity decreases with an increase in temperature, the same is true for water. The following table provides the dynamic viscosity of water with respect to various temperatures.
| Temperature (°C) | Viscosity (cP or mPa·s) |
|---|---|
| 10 | 1.3059 |
| 20 | 1.0016 |
| 30 | 0.79722 |
| 50 | 0.54652 |
| 70 | 0.40355 |
| 90 | 0.31417 |
| 100 | 0.2822 |
Kinematic Viscosity of water
The kinematic viscosity of water can easily be obtained by dividing the above dynamic viscosity values by the water density. Table 2 below provides the kinematic viscosity of water.
| Temperature (°C) | Kinematic Viscosity (m2/s X 10-6) |
| 10 | 1.3059 |
| 20 | 1.004 |
| 30 | 0.801 |
| 50 | 0.553 |
| 70 | 0.413 |
| 90 | 0.326 |
| 100 | 0.294 |
Viscosity of Some Common Substances
The following table states the viscosity of some popular substances.
| Substance | Temperature (°C) | Viscosity (mPa·s) |
| Benzene | 25 | 0.604 |
| Air | 25 | 18.5×10-3 |
| Mercury | 25 | 1.526 |
| Whole milk | 20 | 2.12 |
| Dark beer | 20 | 2.53 |
| Olive oil | 26 | 56.2 |
| Honey | 20 | 2,000–10,000 |
| Ketchup | 25 | 5,000–20,000 |
| Peanut butter | – | 104–106 |
| Pitch | 10–30 (variable) | 2.3×1011 |
Newton’s law of Viscosity
The relationship between the shear stress and the shear rate of fluid under mechanical stress is established by Newton’s law of viscosity. For a given temperature and pressure, Newton’s viscosity law states that the shear stress between two adjacent layers in a fluid is proportional to the velocity gradients between those layers. In another way, it can be stated that the ratio of shear stress to shear rate in a fluid is a constant, and is defined as the coefficient of viscosity. Newtonian fluids obey Newton’s law of viscosity. Non-Newtonian fluids do not follow Newton’s law of viscosity and hence their viscosity varies and is dependent on the shear rate. Dynamic viscosity is the coefficient of viscosity as defined in Newton’s law of viscosity. Equation 1 mentioned above is basically a mathematical representation of Newton’s law of viscosity.
Practical Applications of Viscosity
The concept of Viscosity is used widely in science and technology. The following examples can easily substantiate the applications of viscosity:
- The molecular weight of organic liquids is determined using the knowledge of viscosity.
- In lubrication engineering viscosity data and its variation with temperature is an absolute necessity to decide suitable lubrication for specific equipment. For example, light machines use low viscous liquids whereas highly viscous oils are used in heavy machines.
- For preparing various medicines like syrups viscosity data is required.
- Cooking oils, fats, butter, etc are manufactured to provide a specific viscosity.
- Gums, coolants, petrol as a cleaner, brake fluid, cosmetics, food products, etc all require viscosity data during production to work smoothly.
- Blood circulation inside our body depends on the viscosity of blood.
- In the cosmetic industry, the texture of lotions and creams is largely determined by their viscosity.
Other applications of viscosity can be described as follows:
Viscosity has vast implications across various industries, impacting everything from manufacturing to healthcare.
Industrial Applications
In manufacturing, understanding the viscosity of materials is crucial for processes like mixing, coating, and pumping. For example, in the paint industry, the viscosity of the paint affects application methods and drying times.
Biological Applications
In biology, the viscosity of blood is a vital parameter that influences circulation and oxygen transport. Abnormal viscosity can indicate medical conditions, making its measurement essential in clinical settings.
Environmental Science
In environmental science, viscosity plays a role in the movement of pollutants in water and soil. Understanding how viscosity affects dispersion and degradation can inform remediation efforts.
Food Science
In the food industry, viscosity impacts the texture and stability of products like sauces, dressings, and emulsions. Quality control measures often include viscosity testing to ensure product consistency.
Frequently Asked Questions-Viscosity
What is viscosity?
Viscosity is a measure of a fluid’s resistance to flow. It describes how thick or sticky a fluid is, influencing how easily it can be poured or spread.
What does high viscosity mean?
High viscosity usually means the liquid is thicker. The fluid with high viscosity offers greater flow resistance. Let’s describe the term high-viscosity with an example. Among, the two fluids, water, and honey, honey has a high viscosity as compared to water. So, the resistance force against the flow of water will be less as compared to honey.
What are the Types of Viscosity?
Engineering fluid mechanics provides two types of viscosity; Dynamic Viscosity and Kinematic Viscosity.
Is water viscous?
Viscosity is the property of any fluid. Water being a fluid, specifically liquid, is vicious.
How is viscosity important?
Fluid Viscosity is an important parameter for fluids. For food industries, viscosity provides the texture of food. In some situations, high viscosity is considered to be of superior quality as compared to thin liquids.
Which is the most viscous?
Pitch is the most viscous liquid. Glycerine and lubricating oil also have high viscosities.
How is viscosity measured?
Viscosity can be measured using several methods, including:
- Capillary Viscometers: Measure the time it takes for fluid to flow through a narrow tube.
- Rotational Viscometers: Measure the torque required to rotate a spindle in the fluid.
- Falling Sphere Viscometers: Calculate viscosity based on the time a sphere takes to fall through the fluid.
- Oscillatory Viscometers: Assess the fluid’s resistance to oscillatory motion.
What factors affect viscosity?
Viscosity is influenced by several factors:
- Temperature: Generally, higher temperatures decrease the viscosity of liquids.
- Pressure: In most liquids, higher pressure increases viscosity.
- Fluid Composition: The chemical makeup and additives can alter viscosity.
- Shear Rate: In non-Newtonian fluids, viscosity can change with the rate of shear.
How does viscosity relate to everyday life?
Viscosity impacts various daily activities, such as cooking (the thickness of sauces), automotive (engine oil performance), and cosmetics (the texture of creams and lotions).
Can viscosity change with time?
Yes, some fluids can experience changes in viscosity over time due to factors like temperature fluctuations, chemical reactions, or aging.
What is apparent viscosity?
Apparent viscosity is a term often used in non-Newtonian fluids that do not have a constant viscosity. This type of viscosity can vary depending on the shear rate, making it essential for characterizing complex fluids like polymers and biological fluids.
Does viscosity mean thickness?
Viscosity and thickness are related but not synonymous. Viscosity refers to a fluid’s resistance to flow, while thickness (or consistency) describes the physical property of how “thick” or “thin” a fluid appears. For example, honey is both thick and has a high viscosity, meaning it flows slowly. Water, on the other hand, is thin and has low viscosity, flowing easily. In summary, while viscosity can influence how thick a fluid feels, it specifically measures flow resistance rather than just physical thickness.

Thank you very much sir
very important article
very information al thank you
thank u so much sir
very useful for us
Excellent work